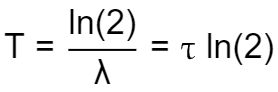Half-Life Calculator
The half-life calculator is a tool that helps you understand the principles of radioactive decay. You can use it to not only learn how to calculate half-life, but also as a way of finding the initial and final quantity of a substance or its decay constant. This article will also present you with the half-life definition and the most common half-life formula.
Half-life definition
Each radioactive material contains stable and unstable nuclei. Stable nuclei don't change, but unstable nuclei undergo a type of radioactive decay, emitting alpha particles, beta particles, or gamma rays and eventually decaying into stable nuclei. Half-life is defined as the time required for half of the unstable nuclei to undergo their decay process.
Each substance has a different half-life. For example, carbon-10 has a half-life of only 19 seconds, making it impossible for this isotope to be encountered in nature. Uranium-233, on the other hand, has a half-life of about 160 000 years.
This term can also be used more generally to describe any kind of exponential decay - for example, the biological half-life of metabolites.
Half-life is a probabilistic measure - it doesn't mean that exactly half of the substance will have decayed after the time of the half-life has elapsed. Nevertheless, it is an approximation that gets very accurate when a sufficient number of nuclei are present.
🙋 One of the applications of knowing half-life is radiocarbon dating. Learn more about that by checking out our Radiocarbon dating calculator.
Half-life formula
We can determine the number of unstable nuclei remaining after time using this equation:
Where:
- – Remaining quantity of a substance after time has elapsed;
- – Initial quantity of this substance; and
- – half-life.
It is also possible to determine the remaining quantity of a substance using a few other parameters:
Where:
- – mean lifetime - the average amount of time a nucleus remains intact; and
- – decay constant (rate of decay).
All three of the parameters characterizing a substance's radioactivity are related in the following way:
How to calculate the half-life
- Determine the initial amount of a substance. For example, .
- Determine the final amount of a substance - for instance, .
- Measure how long it took for that amount of material to decay. In our experiment, we observed that it took 5 minutes.
- Input these values into our half-life calculator. It will compute a result for you instantaneously - in this case, the half-life is equal to .
- If you are not certain that our calculator returned the correct result, you can always check it using the half-life formula.
Confused by exponential formulas? Try our exponent calculator.
Half-life is a similar concept to doubling time in biology. Check our generation time calculator to learn how exponential growth is both useful and a problem in laboratories! Also, we use a similar concept in pharmacology, and we call it the "drug half-life". Find out more about that in our drug half-life calculator.
FAQ
What is half life?
Half-life is defined as the time taken by a substance to lose half of its quantity.
How to calculate half life?
To find half-life:
- Find the substance's decay constant.
- Divide the natural logarithm of 2 or
ln(2)by the decay constant of the substance.
What is the half life of radium?
The half-life of radium-218 is 25.2 x 10-6 seconds.
What is the half life of carbon?
The half-life of carbon-20 is 16 x 10-3 seconds.
What is the half life of uranium?
The half-life of uranium-235 is 22.21 x 1015 seconds.